Our Science and Approach
Neuropore’s drug-discovery efforts are enabled by a robust and efficient research platform comprised of sophisticated computational molecular modeling tools, an unparalleled repertoire of high content imaging, immunohistochemical and biochemical assays of inflammation and autophagy. This research platform is complemented by the extensive utilization of transgenic animal models of neurodegenerative disease that provide translatable biochemical, immunohistochemical and functional measures of the potential therapeutic benefits of candidate compounds.
Neuropore Therapies is pursuing three distinct approaches to address the convergence of protein aggregation, failed protein clearance and neuroinflammation that drive neurodegeneration as depicted by a “triad hypothesis”. These inter-related approaches and mechanisms targeted by Neuropore are summarized below:
•Inhibit the formation of toxic oligomeric protein aggregates of misfolded proteins
•Inhibit brain inflammatory processes involved in disease pathogenesis
•Rectify defects in the cellular mechanisms for clearing aggregates of misfolded proteins
Triad hypothesis incorporates protein dysregulation, inflammation and
autophagy as key components leading to neurodegenerative diseases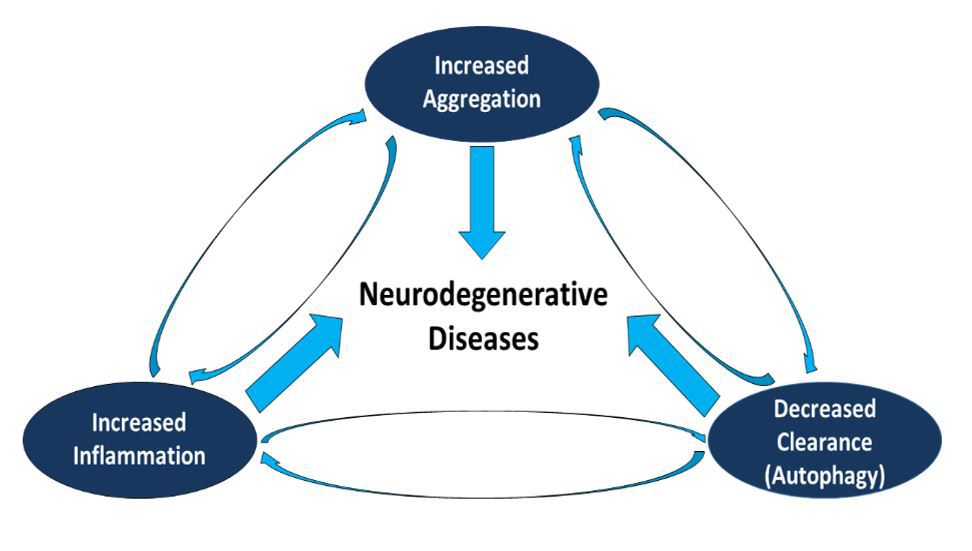
Since the corporate strategy of Neuropore Therapies is to advance therapeutic candidates into early clinical trials for assessments of target engagement and disease-relevant biological activity prior to out-licensing, a strong commitment is also made from the very outset of our drug discovery efforts to the development of clinically translatable markers. These include program-specific markers of target engagement, applicable in early studies in healthy volunteers, and both peripheral and central markers of actions on neuroinflammation applicable to initial studies in patients.
